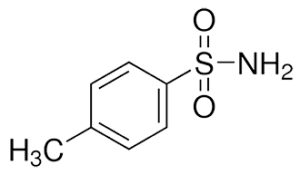
Synonyms
(4-Methylphenyl)sulfonamide; 4-methylbenzenesulfonamidate;
4-Methylbenzenesulfonamide; 4-Sulfamoyltoluene;
4-Methylbenzenesulphonamide; 4-Toluenesulfanamide;
4-Toluenesulfonic acid, amide; N-tosyl amine; N-tosylamine;
para-Toluenesulfonamide p-Methylbenzenesulfonamide;
p-toluene sulfonamide; p-Toluenesulfamide; p-Toluenesulfanamide;
p-Toluenesulfonylamide p-Toluenesulphonamide; p-toluensulfonamide;
p-Tolylsulfonamide; Tolouen-4-sulfonamide; Toluene-4-sulfonamide;
Toluene-4-sulphonamide; toluene-p-sulfonamide; Toluene-p-sulphonamide;
Toluenesulfonamide; toluenesulphonamide; Tolylsulfonamide
Technical Data
CAS Number: 70-55-3
Molecular Formula: C7H9NO2S
Molecular Weight: 171.22
Beilstein Registry Number: 472689
EC Number 200-741-1
FabriChem Specifications
| ITEMS | SPECIFICATIONS |
|---|---|
| Appearance | White crystalline |
| Assay | 99.5% Min. |
| Melting Point | 136—140°C |
| Moisture | 0.2% Max. |
| Color (APHA) | 20 Max. |
| o-Toluene Sulfonamide | 0.2% Max. |
| Turbidity | 10 Max. |
| pH | 5.0 — 7.0 |
| Chloride | 0.01% Max. |
| Sulfate | 0.02% Max. |
| Meta Toluene Sulfonamide | 0.1% Max. |
| Heavy Metal | 20 ppm Max |