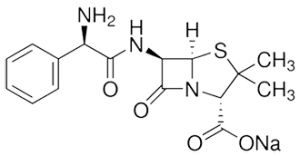
Synonyms
d-(−)-α-aminobenzylpenicillin sodium salt, ampicillin sodium salt
Technical Data
CAS Number: 69-52-3
Molecular Formula: C16H18N3NaO4S
Molecular Weight: 371.39
Beilstein Registry Number: 4119211
EC Number: 200-708-1
Ampicillin Sodium Sterile Crystalline (EP Grade)
| ITEMS | SPECIFICATIONS |
|---|---|
| Description | A white or almost white powder, hygroscopic |
| Solubility | Freely soluble in water, practically insoluble in acetone, fatty oils and liquid paraffin. |
| Identification (IR) | The IR spectrum should be concordant with that of Ampicillin trihydrate working standard. |
| Identification (TLC) | The principal spot in the chromatogram obtained with the test solution should be similar in position, color and size to the principal spot in the chromatogram obtained with the reference solution (a) |
| Test for Penicillins | A dark yellow color should develop |
| Test for Sodium | A dense, white precipitate should form, indicating the presence of sodium |
| Appearance of Solution (10% w/v solution) | |
| Clarity (1.0 g of sample dissolved in 10 ml of 1 M Hydrochloric acid or 10 ml of water) | Not more opalescent than reference suspension-II. |
| Absorbance at 430 nm (1.0 g of sample dissolved in 10 ml of water) | NMT 0.15 |
| pH (10%w/v in carbon dioxide-free water) | 8.0 to 10.0 |
| Specific Optical Rotation (0.25g of substance in 100 ml 0.4% w/v solution of potassium hydrogen phthalate, at 25°C) | Between +258° and +287° (Calculated on anhydrous basis) |
| Ampicillin Dimer (by HPLC, %w/w) | NMT 4.5% |
| Any Individual Impurity (by HPLC, %w/w) | NMT 2.0% |
| Methylene Chloride (by GC, %w/w) | NMT 0.2% |
| Heavy Metals | NMT 20 ppm |
| Water (by KF, %w/w, determined on 0.300g) | NMT 2.0% |
| Sterility | Should be sterile |
| Bacterial Endotoxins (IU/mg of Ampicillin) | NMT 0.15 IU/mg |
| Assay (by HPLC, % w/w) As Ampicillin Sodium (calculated on anhydrous basis) | 91.0% - 102.0% |
| Residual Solvents: | NMT 410 ppm |
| Acetonitrile (by GC) | NMT 1.0 ppm |
| Sodium Thiocyanate (by UV, w/w) | |
| Particulate Matter (particles per gram) | |
| Visual Inspection | Should be free from visible particles |
| Particulate Matter (by LBPC) a) ≥ 10 µ b) ≥ 25 µ | NMT 6,000/g NMT 600/g |
| Untapped Density | Report Only |
| Tapped Density | Report Only |
Ampicillin Sodium Sterile Crystalline (USP Grade)
| ITEMS | SPECIFICATIONS |
|---|---|
| Description | White to off-white, odorless or practically odorless, crystalline powder; hygroscopic. |
| Solubility | chloride and dextrose solutions. |
| Identification a) by IR | The IR spectrum should be concordant with that of Ampicillin sodium working standard. |
| b) Test for Sodium | A dense, white precipitate should form, indicating presence of sodium ions. |
| Crystallinity | The particles should show birefringence and exhibit extinction positions. |
| pH (1% w/v in carbon dioxide-free water) | 8.0 to 10.0 |
| Water (K.F.) | NMT 2.0% w/w |
| Sterility Test | Should be Sterile |
| Bacterial Endotoxins | NMT 0.15 USP EU/mg |
| Assay (HPLC) (calculated on anhydrous basis) | 845 μg/mg to 988 μg/mg |
| Particulate Matter | Should be free from visible particles |
| Particulate Matter (by LBPC) a) ≥ 10 µ b) ≥ 25 µ | NMT 6,000/g NMT 600/g |
| Untapped Density | Report Only |
| Tapped Density | Report Only |
| Residual Solvents (by GC) Acetonitrile Methylene chloride | NMT 410 ppm NMT 2000 ppm |